|
2/22/2023 0 Comments Goldstein cathode ray experiment 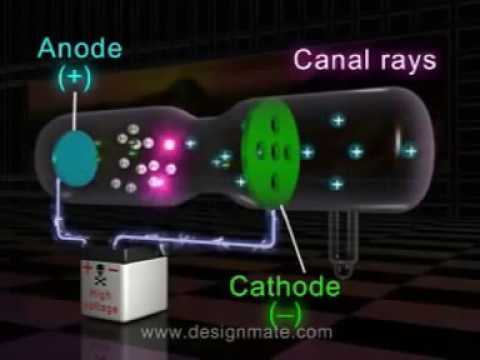
The first subatomic particle to be discovered was the electron. Why was the first subatomic particle discovered?Įlectrons. How was Proton Discovered? In gold foil experiment, Rutherford bombarded alpha particles on an ultrathin gold foil and then detected the scattered alpha particles on zinc sulphide (ZnS) screen. In 1909, Rutherford discovered proton in his gold foil experiment. 
Which experiment led to the discovery of protons and how? One plate is connected to positive terminal of high voltage power supply and the other to negative terminal. It is made of a glass tube which consists of two metallic plates. What is discharge tube explain with diagram?ĭischarge tube is also called “CROOCK TUBE”. Thomson studied the characteristics and the constituents of cathode rays. Who performed discharge tube experiment?Įxperiments on the discharge tube were first performed in 1878 by an English physicist, William Crookes. Based on the conclusions drawn from the gold-foil experiment, Rutherford is also credited with the discovery of the atomic nucleus. a proton) is present in the nuclei of all other atoms in the year 1917. The discovery of the proton is credited to Ernest Rutherford, who proved that the nucleus of the hydrogen atom (i.e. What is discovered by Goldstein?Įugen Goldstein/Inventions Who discovered the proton and with what experiment? Due to passage of electric current, a stream of rays is passed in the tube originating from cathode. In discharge tube experiment, at low pressure and at very high voltage, an electric current is passed. The charge of an electron was measured by R. Thomson in Cathode Ray Tube (CRT) experiment. What was the experiment that led to the discovery of the electron?Įlectron was discovered by J. The discovery of X-rays When the voltage applied to a Crookes tube is high enough, around 5,000 volts or greater, it can accelerate the electrons to a high enough velocity to create X-rays when they hit the anode or the glass wall of the tube. What did scientists discover with discharge tubes? These protons add the overall positive charge of an atom. Thomson also determined e/m, ratio of the charge e to the mass m of the material particle which constituted these rays. Thomson, using a cathode rays while doing discharge tube experiments. The electron was discovered in 1896, by the British physicist J. … How was the presence of electron and proton in the atom discovered in the discharge tube experiment? He found a green glow on the wall of his glass tube and attributed it to rays emanating from the cathode. Their work culminated in the discovery by English physicist J.J. How electrons are discovered with the help of discharge tube?ĭuring the 1880s and ’90s scientists searched cathode rays for the carrier of the electrical properties in matter. 
10 What did JJ Thomson discover about the atom?.9 What was the cathode ray tube experiment?.8 Who discovered electric discharge in gases?.7 Why was the first subatomic particle discovered?.6 Who performed discharge tube experiment?.5 Who discovered the proton and with what experiment?.3 What did scientists discover with discharge tubes?.2 How was the presence of electron and proton in the atom discovered in the discharge tube experiment?.1 How electrons are discovered with the help of discharge tube?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
